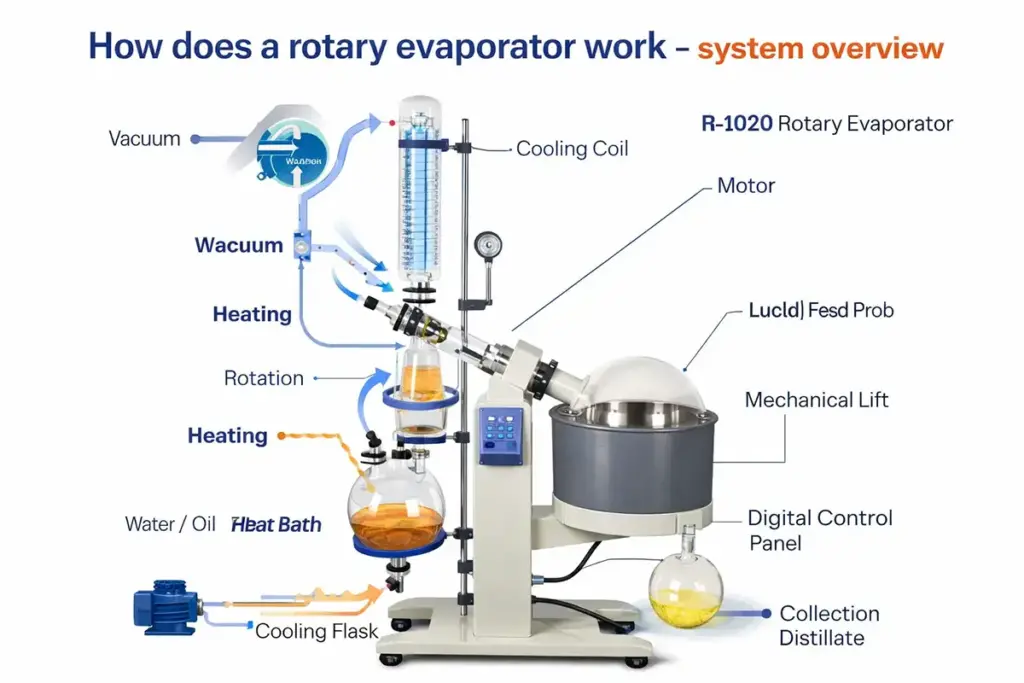
A rotary evaporator (“rotovap”) looks like a simple lab appliance, but its performance comes from a carefully balanced trio: vacuum (to lower boiling points), rotation (to create a thin film), and condensation (to capture vapor efficiently). This guide explains the real working principle in plain language—and why each part matters when choosing a system.
1) The Big Picture: What a Rotovap Actually Does
The goal of a rotary evaporator is straightforward: remove a volatile solvent from a mixture (for example, concentrating an extract or recovering a solvent) while keeping temperatures as gentle as possible.
The machine achieves this by heating the sample in a bath, spinning the evaporation flask, pulling a vacuum to reduce pressure, and then condensing the solvent vapor into a receiving flask. The “magic” is not one feature—it’s how the three features cooperate.
Why it’s so widely used
In many labs and pilot-scale processes, rotovaps provide a practical balance of speed, solvent recovery, and product protection—especially compared with open-beaker evaporation or simple distillation setups.
2) Vacuum: Lower Pressure, Lower Boiling Point (Less Heat Damage)
If one question sits at the center of how does a rotary evaporator work, it’s this: Why apply vacuum at all? Vacuum reduces the pressure above the liquid. When pressure drops, the solvent can boil at a lower temperature. That means faster evaporation without needing to “cook” sensitive compounds.
If vacuum is already applied, why keep the water (or oil) bath hot?
Because vacuum lowers the boiling point, but it does not eliminate the need for energy. The bath provides controlled heat to supply the latent heat of vaporization. In practice, a moderate bath temperature plus stable vacuum typically evaporates faster than “very hot bath, weak vacuum.”
Practical benchmark: many laboratory rotovaps specify a maximum vacuum in the range of <133 Pa (≈1.33 mbar, ≈1 Torr). At these pressures, common solvents can boil dramatically below their normal boiling points, helping protect heat-sensitive samples.
Authoritative data point: According to NIST (National Institute of Standards and Technology) reference principles on phase equilibria, a liquid’s boiling point decreases as external pressure decreases—this pressure–temperature dependence is the physical basis of vacuum evaporation.
3) Thin Film (Rotation): More Surface Area, Less Bumping, Faster Evaporation
Rotation is not for show. When the evaporation flask spins, the liquid spreads into a thin film over the inner wall of the flask. A thin film evaporates faster because the solvent has a larger surface area exposed to the reduced-pressure environment.

Rotation also helps reduce bumping (sudden violent boiling). A more even film and continuous mixing produce steadier boiling, which is important for yield (less sample loss) and cleanliness (fewer splashes into the condenser).
Does higher RPM always mean faster evaporation?
Not always. Higher RPM can improve film formation up to a point, but excessive speed may cause unstable films, foaming, or inefficient heat transfer. In real operation, the “best” speed is the one that creates a stable, even film with controlled boiling—often within typical ranges such as 0–120 rpm on many lab units.
Typical configurations include stepless speed regulation and working ranges like 0–120 rpm (common on smaller models), allowing fine control for different viscosities and solvent behaviors.
4) Condensation: Turning Vapor Back Into Liquid (Solvent Recovery)
After vacuum and thin-film evaporation generate solvent vapor, the condenser’s job is to capture that vapor before it reaches the vacuum pump. Efficient condensation improves recovery, keeps vacuum stable, and reduces emissions.
Many systems use a vertical double condenser or a vertical double-coil condenser to increase cooling surface area. The better the condenser performance, the lower the solvent load on the pump—and the smoother the overall operation.

Why can weak condensation make evaporation look “slow” even with strong vacuum?
Because vapor must have somewhere to go. If condensation is insufficient, vapor accumulates, pressure rises, and effective evaporation slows. In other words, vacuum quality and condenser capacity must match; improving only one side can still bottleneck the system.
Authoritative data point: The U.S. EPA’s solvent management guidance emphasizes that efficient condensation and capture reduce solvent losses and emissions in recovery processes, reinforcing why condenser performance is not optional in solvent evaporation workflows.
5) Step-by-Step Workflow (Simple Mental Model)
Here is the easiest way to remember the working principle:
- Heat adds energy gently (water/oil bath).
- Vacuum lowers boiling points so solvents evaporate sooner.
- Rotation spreads liquid into a thin film for faster mass transfer and steadier boiling.
- Condenser cools vapor back to liquid for clean collection in the receiving flask.
Practical takeaway
When troubleshooting a “slow rotovap,” it is usually one of these: vacuum leaks, insufficient cooling, or unstable boiling due to poor film formation (speed/temperature mismatch).
6) Real Parameters: What to Look for When Choosing a Rotary Evaporator
Specs matter because they map directly to the three core functions (vacuum, thin film, condensation) and to throughput. Below are example parameter ranges from common lab and pilot-scale configurations.
| Example Models | Evaporating Flask | Speed Range | Max Vacuum | Evaporation Rate (H₂O) | Condenser Type |
|---|---|---|---|---|---|
| RE-201D / RE-301 / RE-501 | 2–5 L | 0–120 rpm | <133 Pa (≈1.33 mbar) | >1 to >1.5 L/h | (Typical lab condenser) |
| RE-1002 / RE-2002 / RE-5002 | 10–50 L | 0–120 rpm (some 0–90) | (System-dependent) | >3 / >5 / >9 L/h | Vertical Double Condenser |
| R1005 / R1010 / R1020 / R1050 | 5 / 10 / 20 / 50 L | 10–140 or 20–120 rpm | (System-dependent) | (Application-dependent) | Vertical double coil / high-efficiency reflux types |
Mobile note: the table is horizontally scrollable on small screens to preserve readability and prevent layout breaking.
If evaporation speed is the priority, pay attention to evaporation rate, condenser design, bath power, and vacuum stability. If sample protection is the priority, focus on stable deep vacuum, good sealing (e.g., PTFE + fluoro rubber), and controlled temperature resolution.
7) Related Equipment and Next Steps
Rotary evaporation is often part of a broader solvent recovery or purification workflow. For deeper product details and adjacent solutions:
- Explore a product overview and options on the rotary evaporator page.
- For larger-scale recovery discussions, see: solvent recovery distillation units guide .
- If the process calls for alternative gentle separation at higher purity, consider: short path distillation unit .
Selection shortcut
Match capacity (flask size) to batch volume, then ensure the condenser and vacuum system can handle the expected solvent load. That alignment is what makes a rotovap feel “fast” and “easy” in daily use.
Summary: The Working Principle in One Sentence
A rotary evaporator works by reducing pressure (vacuum) to lower the solvent’s boiling point, spinning the flask to form a thin film for rapid, stable evaporation, and condensing the vapor to recover solvent efficiently—delivering faster concentration at gentler temperatures.
